Lyo-Ready™ LAMP/RT-LAMP Mixes
Meridian’s Lyo-Ready™ LAMP/RT-LAMP Mixes are ready-to-use, 4x concentrated, fully optimized mixes for isothermal amplification methods such as Loop mediated isothermal amplification (LAMP), designed to dramatically speed up assay development time and reduce time to results.
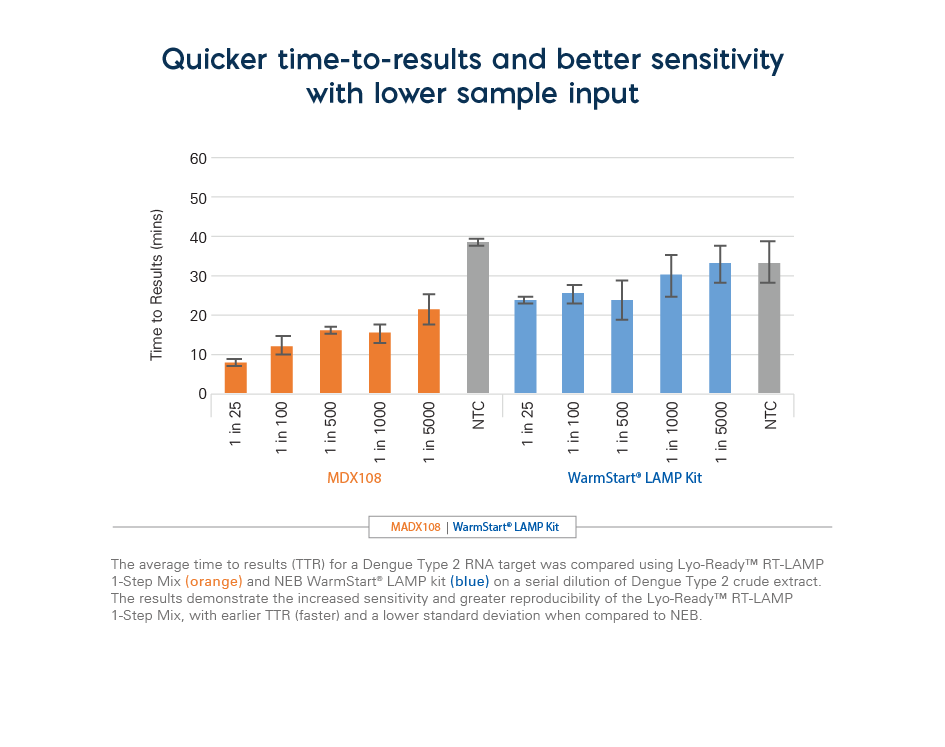
Loop mediated isothermal amplification is a highly sensitive nucleic acid amplification method that has gained popularity in recent years due to its advantages over qPCR, such as quicker time to results, simple assay workflow, and point-of-care usability. However, the initial assay design and development can be more complex with 4 to 6 primers which are used in combination to generate a robust, fast, and sensitive result. Through extensive R&D research, Meridian has discovered a blend of bst DNA polymerase, reverse transcriptase, dNTPs, Mg2+, excipients, and stabilizers that are used with customer-selected primers and probes for creating any custom DNA or RNA LAMP test. Experimental data on both RNA and DNA templates demonstrate that the mixes are up to 2x faster than other suppliers, generating results in half the time. In addition, the mixes incorporate Meridian’s Lyo-Ready™ technology and can be used in a liquid format or lyophilized to create an ambient-temperature stable assay (please see the product guide and FAQs for recommendations on lyophilization parameters).
By using a ready-to-use master mix such as Lyo-Ready™ LAMP Mix or Lyo-Ready™ RT-LAMP Mix, LAMP test assay development is faster, simpler, and more efficient, speeding up your product development time and allowing you to commercialize your new products faster.
Can also be used wet
Have questions about a product?
Contact us to learn more about Meridian’s molecular or immunoassay reagent portfolio. We want to hear from you!
Lyo-Ready™ LAMP Mix, MDX097
Available in 5 mL (800 Rxns) or 50 mL (8,000 Rxns) aliquots
Lyo-Ready™ RT-LAMP 1-Step Mix, MDX108
Available in 5 mL (800 Rxns) or 50 mL (8,000 Rxns) aliquots
The Lyo-Ready™ LAMP Mixes are 4x master mix formulated with excipients optimized for freeze-drying to create ambient-temperature stable assays. Given cold chain management is a major limiting factor in resource-limited settings, ambient-temperature stable point-of-care tests (POCT) offer several advantages, including no refrigeration, reduced shipping, and handling costs, and extended shelf-life. By using Meridian’s Lyo-Ready™ LAMP Mixes, the process of developing a sensitive and high-performing assay is made significantly easier, with the added flexibility of using the mix in a wet or dried-down format.
Catalogs & Brochures
Master Mixes for Molecular Ambient-Temperature Stable AssaysLyo-ReadyTM LAMP Mix, 4x
Master Mixes for Molecular Ambient-Temperature Stable AssaysLyo-Ready RT-LAMP 1-Step Mix, 4x
FAQs: Lyo-Ready™ LAMP Mixes
The main difference between loop mediated isothermal amplification and qPCR is that in loop mediated isothermal amplification, the amplification is achieved using a single constant temperature, while in the qPCR, amplification requires cycling between multiple temperatures on a thermocycler. Instead of melting DNA strands apart at high temperatures, loop mediated isothermal amplification uses polymerases with high strand displacement activity, like Bst DNA polymerases. As these polymerases do not need multiple temperatures, they can amplify a target in less than an hour, and in some cases in as little time as 10 minutes.
Lyo-Ready™ LAMP and Lyo-Ready™ RT-LAMP 1-Step Mix can both be used directly as a liquid mix, or lyophilized, without changing the sensitivity of the assay. The master mixes are 4x concentration, this means that in a 20 L reaction, 5 L the master mix and 1 L of primers and probes are used allowing for up to 14 L of sample to be added, however if the master mix, primers, and probes are lyophilized, then up to 20 L of sample can be added.
Yes, it is possible to designed a multiplex LAMP test using different primers and probes, however they can be difficult to design and optimize as each sequence requires three sets of primers.
Yes, the Lyo-Ready™ LAMP and Lyo-Ready™ RT-LAMP 1-Step Mix can be used as a liquid mix or lyophilized and stored for up to 24 months without needing to change the reaction conditions and this drying/ambient temperature storage will not affect the sensitivity of the POCT.
Get In Touch With A Specialist
Have questions about a product? Want to learn more about Meridian’s molecular or immunoassay reagent portfolio? We want to hear from you!